Why Can an Enzyme Be Used Again and Again
A fundamental task of proteins is to act as enzymes—catalysts that increment the rate of almost all the chemical reactions within cells. Although RNAs are capable of catalyzing some reactions, most biological reactions are catalyzed by proteins. In the absence of enzymatic catalysis, almost biochemical reactions are so slow that they would non occur nether the mild conditions of temperature and pressure that are uniform with life. Enzymes accelerate the rates of such reactions by well over a meg-fold, so reactions that would take years in the absenteeism of catalysis can occur in fractions of seconds if catalyzed by the appropriate enzyme. Cells contain thousands of different enzymes, and their activities determine which of the many possible chemical reactions actually take place within the cell.
The Catalytic Activity of Enzymes
Similar all other catalysts, enzymes are characterized past two primal properties. First, they increase the charge per unit of chemical reactions without themselves being consumed or permanently altered by the reaction. 2d, they increase reaction rates without altering the chemical equilibrium between reactants and products.
These principles of enzymatic catalysis are illustrated in the following instance, in which a molecule acted upon by an enzyme (referred to as a substrate [S]) is converted to a production (P) as the result of the reaction. In the absenteeism of the enzyme, the reaction can be written as follows:
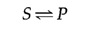
The chemic equilibrium between South and P is determined past the laws of thermodynamics (as discussed further in the next section of this chapter) and is represented past the ratio of the forrad and reverse reaction rates (S→P and P→S, respectively). In the presence of the advisable enzyme, the conversion of S to P is accelerated, only the equilibrium betwixt South and P is unaltered. Therefore, the enzyme must accelerate both the forward and reverse reactions as. The reaction tin be written as follows:
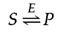
Note that the enzyme (Due east) is not altered by the reaction, so the chemical equilibrium remains unchanged, determined solely by the thermodynamic backdrop of S and P.
The effect of the enzyme on such a reaction is all-time illustrated past the energy changes that must occur during the conversion of S to P (Effigy 2.22). The equilibrium of the reaction is determined by the last free energy states of S and P, which are unaffected past enzymatic catalysis. In order for the reaction to proceed, withal, the substrate must get-go exist converted to a higher energy state, called the transition state. The free energy required to attain the transition state (the activation free energy) constitutes a barrier to the progress of the reaction, limiting the rate of the reaction. Enzymes (and other catalysts) human activity by reducing the activation energy, thereby increasing the rate of reaction. The increased charge per unit is the same in both the forward and reverse directions, since both must pass through the same transition state.
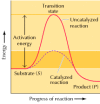
Figure 2.22
Energy diagrams for catalyzed and uncatalyzed reactions. The reaction illustrated is the unproblematic conversion of a substrate S to a product P. Considering the final energy state of P is lower than that of S, the reaction proceeds from left to correct. For the (more...)
The catalytic activity of enzymes involves the binding of their substrates to class an enzyme-substrate circuitous (ES). The substrate binds to a specific region of the enzyme, called the active site. While bound to the agile site, the substrate is converted into the product of the reaction, which is so released from the enzyme. The enzyme-catalyzed reaction tin can thus exist written as follows:
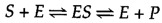
Note that E appears unaltered on both sides of the equation, then the equilibrium is unaffected. All the same, the enzyme provides a surface upon which the reactions converting South to P tin can occur more than readily. This is a consequence of interactions between the enzyme and substrate that lower the energy of activation and favor germination of the transition land.
Mechanisms of Enzymatic Catalysis
The binding of a substrate to the agile site of an enzyme is a very specific interaction. Active sites are clefts or grooves on the surface of an enzyme, usually composed of amino acids from dissimilar parts of the polypeptide chain that are brought together in the third structure of the folded protein. Substrates initially bind to the active site past noncovalent interactions, including hydrogen bonds, ionic bonds, and hydrophobic interactions. Once a substrate is bound to the active site of an enzyme, multiple mechanisms tin can accelerate its conversion to the product of the reaction.
Although the simple instance discussed in the previous section involved merely a single substrate molecule, virtually biochemical reactions involve interactions between two or more different substrates. For case, the formation of a peptide bond involves the joining of ii amino acids. For such reactions, the binding of two or more substrates to the active site in the proper position and orientation accelerates the reaction (Figure 2.23). The enzyme provides a template upon which the reactants are brought together and properly oriented to favor the germination of the transition state in which they interact.

Figure 2.23
Enzymatic catalysis of a reaction betwixt ii substrates. The enzyme provides a template upon which the 2 substrates are brought together in the proper position and orientation to react with each other.
Enzymes accelerate reactions also by altering the conformation of their substrates to arroyo that of the transition country. The simplest model of enzyme-substrate interaction is the lock-and-central model, in which the substrate fits precisely into the active site (Figure 2.24). In many cases, even so, the configurations of both the enzyme and substrate are modified past substrate binding—a process called induced fit. In such cases the conformation of the substrate is altered so that information technology more closely resembles that of the transition land. The stress produced past such distortion of the substrate tin further facilitate its conversion to the transition land by weakening critical bonds. Moreover, the transition land is stabilized past its tight binding to the enzyme, thereby lowering the required energy of activation.

Effigy 2.24
Models of enzyme-substrate interaction. (A) In the lock-and-key model, the substrate fits precisely into the active site of the enzyme. (B) In the induced-fit model, substrate bounden distorts the conformations of both substrate and enzyme. This baloney (more than...)
In addition to bringing multiple substrates together and distorting the conformation of substrates to approach the transition land, many enzymes participate directly in the catalytic process. In such cases, specific amino acrid side chains in the active site may react with the substrate and form bonds with reaction intermediates. The acidic and basic amino acids are frequently involved in these catalytic mechanisms, as illustrated in the following discussion of chymotrypsin as an instance of enzymatic catalysis.
Chymotrypsin is a member of a family of enzymes (serine proteases) that assimilate proteins by catalyzing the hydrolysis of peptide bonds. The reaction can be written as follows:

The different members of the serine protease family (including chymotrypsin, trypsin, elastase, and thrombin) have distinct substrate specificities; they preferentially carve peptide bonds adjacent to dissimilar amino acids. For case, whereas chymotrypsin digests bonds adjacent to hydrophobic amino acids, such as tryptophan and phenylalanine, trypsin digests bonds next to bones amino acids, such as lysine and arginine. All the serine proteases, however, are similar in structure and use the aforementioned machinery of catalysis. The active sites of these enzymes incorporate three critical amino acids—serine, histidine, and aspartate—that drive hydrolysis of the peptide bond. Indeed, these enzymes are called serine proteases because of the central function of the serine residue.
Substrates bind to the serine proteases past insertion of the amino acid adjacent to the cleavage site into a pocket at the active site of the enzyme (Figure 2.25). The nature of this pocket determines the substrate specificity of the different members of the serine protease family. For instance, the bounden pocket of chymotrypsin contains hydrophobic amino acids that interact with the hydrophobic side chains of its preferred substrates. In dissimilarity, the binding pocket of trypsin contains a negatively charged acidic amino acid (aspartate), which is able to form an ionic bond with the lysine or arginine residues of its substrates.
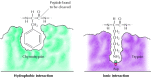
Effigy 2.25
Substrate binding by serine proteases. The amino acid next to the peptide bond to be cleaved is inserted into a pocket at the agile site of the enzyme. In chymotrypsin, the pocket binds hydrophobic amino acids; the bounden pocket of trypsin contains (more...)
Substrate bounden positions the peptide bond to be cleaved adjacent to the active site serine (Figure 2.26). The proton of this serine is and so transferred to the active site histidine. The conformation of the active site favors this proton transfer because the histidine interacts with the negatively charged aspartate residue. The serine reacts with the substrate, forming a tetrahedral transition state. The peptide bond is and so cleaved, and the C-terminal portion of the substrate is released from the enzyme. However, the Northward-terminal peptide remains bound to serine. This state of affairs is resolved when a water molecule (the second substrate) enters the active site and reverses the preceding reactions. The proton of the water molecule is transferred to histidine, and its hydroxyl grouping is transferred to the peptide, forming a second tetrahedral transition state. The proton is then transferred from histidine back to serine, and the peptide is released from the enzyme, completing the reaction.

Figure 2.26
Catalytic mechanism of chymotrypsin. Three amino acids at the active site (Ser-195, His-57, and Asp-102) play critical roles in catalysis.
This example illustrates several features of enzymatic catalysis; the specificity of enzyme-substrate interactions, the positioning of dissimilar substrate molecules in the active site, and the involvement of active-site residues in the formation and stabilization of the transition land. Although the thousands of enzymes in cells catalyze many different types of chemical reactions, the same basic principles apply to their operation.
Coenzymes
In addition to binding their substrates, the active sites of many enzymes bind other small molecules that participate in catalysis. Prosthetic groups are small molecules bound to proteins in which they play critical functional roles. For example, the oxygen carried past myoglobin and hemoglobin is bound to heme, a prosthetic group of these proteins. In many cases metal ions (such every bit zinc or atomic number 26) are bound to enzymes and play central roles in the catalytic process. In addition, diverse low-molecular-weight organic molecules participate in specific types of enzymatic reactions. These molecules are called coenzymes because they work together with enzymes to heighten reaction rates. In contrast to substrates, coenzymes are non irreversibly altered by the reactions in which they are involved. Rather, they are recycled and tin participate in multiple enzymatic reactions.
Coenzymes serve every bit carriers of several types of chemic groups. A prominent example of a coenzyme is nicotinamide adenine dinucleotide (NAD +), which functions as a carrier of electrons in oxidation-reduction reactions (Figure two.27). NAD+ tin can accept a hydrogen ion (H+) and two electrons (due east-) from one substrate, forming NADH. NADH can so donate these electrons to a 2d substrate, re-forming NAD+. Thus, NAD+ transfers electrons from the first substrate (which becomes oxidized) to the 2d (which becomes reduced).

Figure ii.27
Office of NAD+ in oxidation-reduction reactions. (A) Nicotinamide adenine dinucleotide (NAD+) acts as a carrier of electrons in oxidation-reduction reactions by accepting electrons (e-) to form NADH. (B) For example, NAD+ can accept electrons from one substrate (more...)
Several other coenzymes besides act every bit electron carriers, and still others are involved in the transfer of a variety of additional chemical groups (e.g., carboxyl groups and acyl groups; Table 2.1). The same coenzymes function together with a variety of different enzymes to catalyze the transfer of specific chemic groups between a wide range of substrates. Many coenzymes are closely related to vitamins, which contribute part or all of the construction of the coenzyme. Vitamins are not required by leaner such as Eastward. coli but are necessary components of the diets of homo and other higher animals, which have lost the power to synthesize these compounds.
Regulation of Enzyme Action
An important feature of most enzymes is that their activities are not abiding but instead tin be modulated. That is, the activities of enzymes can be regulated so that they function appropriately to meet the varied physiological needs that may arise during the life of the cell.
One common type of enzyme regulation is feedback inhibition, in which the product of a metabolic pathway inhibits the activity of an enzyme involved in its synthesis. For example, the amino acrid isoleucine is synthesized past a series of reactions starting from the amino acrid threonine (Figure 2.28). The starting time footstep in the pathway is catalyzed past the enzyme threonine deaminase, which is inhibited by isoleucine, the end product of the pathway. Thus, an adequate corporeality of isoleucine in the cell inhibits threonine deaminase, blocking further synthesis of isoleucine. If the concentration of isoleucine decreases, feedback inhibition is relieved, threonine deaminase is no longer inhibited, and additional isoleucine is synthesized. By then regulating the activity of threonine deaminase, the jail cell synthesizes the necessary amount of isoleucine but avoids wasting energy on the synthesis of more than isoleucine than is needed.

Effigy ii.28
Feedback inhibition. The showtime step in the conversion of threonine to iso-leucine is catalyzed by the enzyme threonine deaminase. The activity of this enzyme is inhibited by isoleucine, the end product of the pathway.
Feedback inhibition is one case of allosteric regulation, in which enzyme activity is controlled past the binding of small molecules to regulatory sites on the enzyme (Effigy 2.29). The term "allosteric regulation" derives from the fact that the regulatory molecules demark not to the catalytic site, merely to a distinct site on the poly peptide (allo= "other" and steric= "site"). Bounden of the regulatory molecule changes the conformation of the poly peptide, which in turn alters the shape of the agile site and the catalytic activity of the enzyme. In the case of threonine deaminase, bounden of the regulatory molecule (isoleucine) inhibits enzymatic activeness. In other cases regulatory molecules serve as activators, stimulating rather than inhibiting their target enzymes.
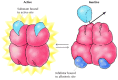
Figure ii.29
Allosteric regulation. In this example, enzyme activity is inhibited by the binding of a regulatory molecule to an allosteric site. In the absence of inhibitor, the substrate binds to the agile site of the enzyme and the reaction proceeds. The binding (more...)
The activities of enzymes can also be regulated by their interactions with other proteins and by covalent modifications, such as the improver of phosphate groups to serine, threonine, or tyrosine residues. Phosphorylation is a particularly common mechanism for regulating enzyme activity; the add-on of phosphate groups either stimulates or inhibits the activities of many different enzymes (Figure ii.thirty). For example, muscle cells respond to epinephrine (adrenaline) by breaking down glycogen into glucose, thereby providing a source of energy for increased muscular activity. The breakdown of glycogen is catalyzed by the enzyme glycogen phosphorylase, which is activated by phosphorylation in response to the bounden of epinephrine to a receptor on the surface of the muscle cell. Poly peptide phosphorylation plays a key role in controlling not simply metabolic reactions but also many other cellular functions, including prison cell growth and differentiation.

Figure 2.xxx
Poly peptide phosphorylation. Some enzymes are regulated by the improver of phosphate groups to the side-chain OH groups of serine (equally shown here), threonine, or tyrosine residues. For example, the enzyme glycogen phosphorylase, which catalyzes the conversion (more...)
Source: https://www.ncbi.nlm.nih.gov/books/NBK9921/
0 Response to "Why Can an Enzyme Be Used Again and Again"
Post a Comment